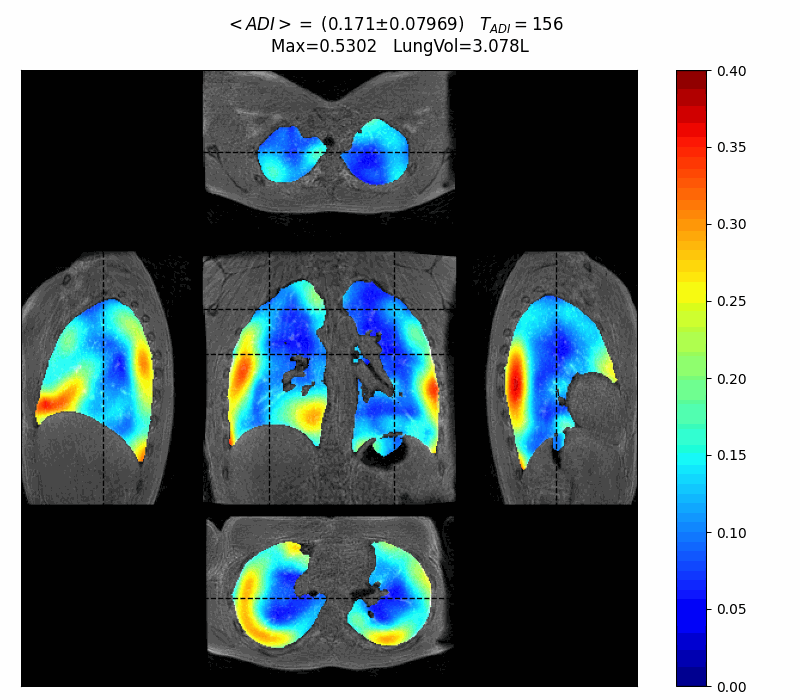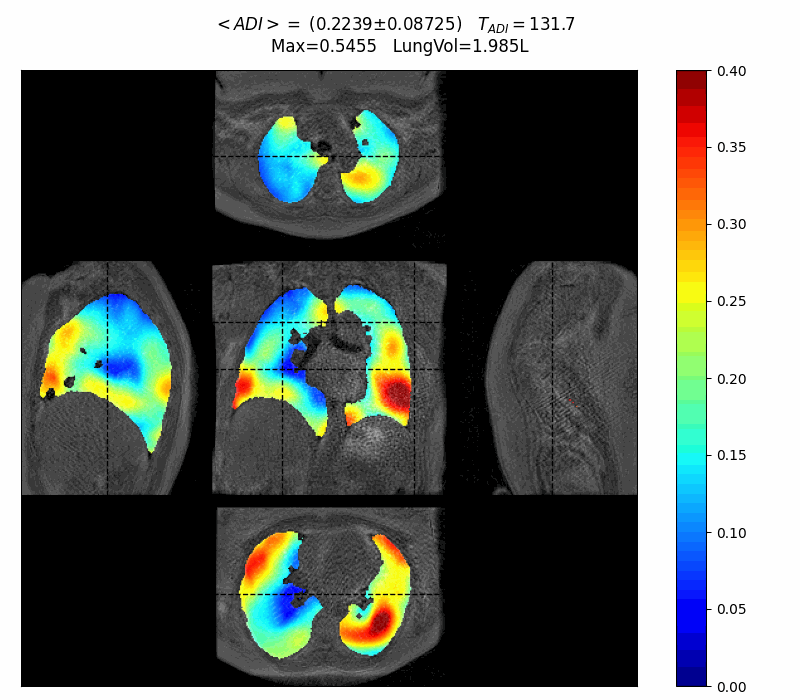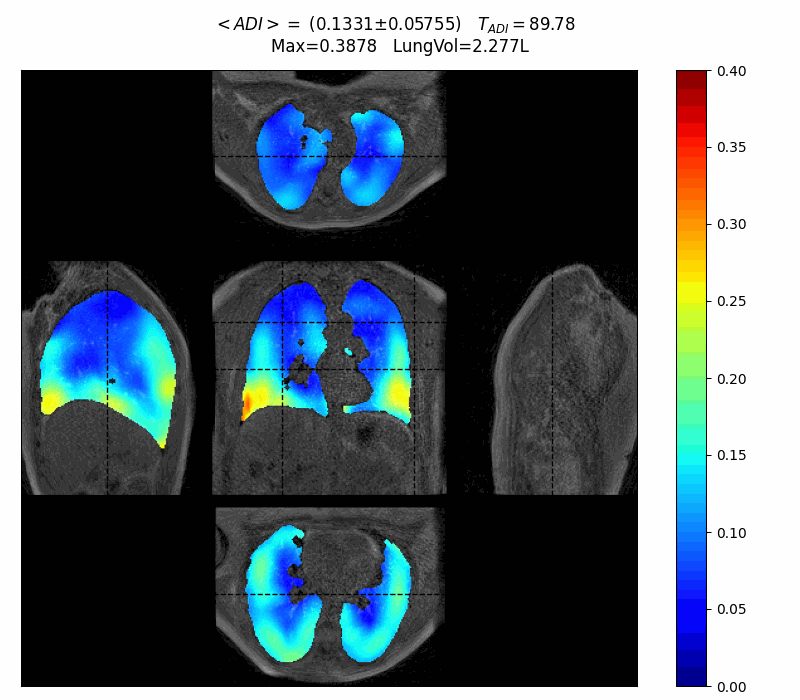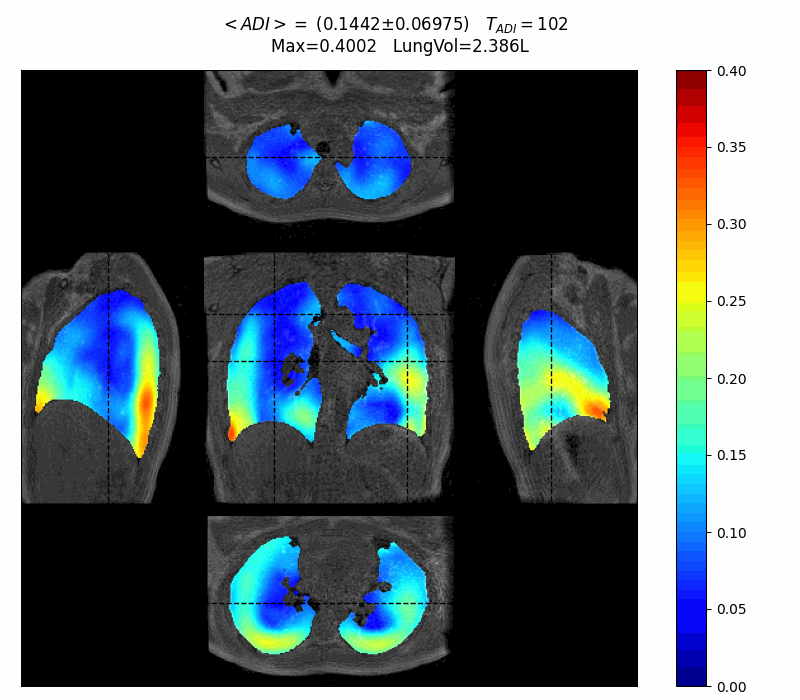
First patient interview
WP4-1 Progress report
A pilot interview about patient experiences during the 3D MRI spirometry protocol was conducted by the social humanistic study group on October 30, 2023 (preliminary phase of WP4-1 “Social humanistic study”).
Why interviewing patients about their experience?
The goal of interviewing patients following their MRI spirometry exam is to investigate how they experience this new protocol. It is indeed well-known that, for the clinical process, patient experiences are very important. When a technique allows to measure what is intended to measure but with a procedure that is uncomfortable, awkward or impractical in its use, compliance does not benefit. Exploring the perspective of the patient is thus aimed at helping design the protocol that will most benefit the patient not only in terms of diagnostic and disease management, but also in terms of personal acceptance.
Why performing conversational interviews?
Conversational interviews are arguably one of the best ways to capture subjective perspectives, experiences and interpretations. It allows interviewees to use their own words, so without being limited by restrictive questionnaires that define relevant topics, questions and answers beforehand. It has been shown in various fields that this type of “qualitative” interview allows for the collection of incredibly rich and detailed data, and that it can inform and complement “quantitative” interviews that are based on well-defined and agreed on questions with limited-choice responses. Conversational interviews have thus been proposed in the social humanistic study plan in order to get insight into patient’s experiences of the study protocol.
The pilot interview we conducted in October 2023 served two purposes:
1. To get a preliminary understanding of possible patient experiences related to the protocol
2. To get feedback on the topic list and structure of the interview with the test subject, so as to verify their relevance and comprehensiveness.
Conduct of the interview
The pilot interview was conducted online by Irene Groenevelt (WP4, post-doc researcher from Tilburg University trained in qualitative research and ethnography) in the presence of Claire Barakat (WP1 and WP4, PhD researcher from Inserm who will help with patient interviewing at French sites later on in the process).
The interview started with an oral explanation of the purpose of the interview. The interviewee was then reminded of the elements of the informed consent form, in particular of the way that the data will be handled (i.e. safe data storage, anonymization) and of the fact that she can withdraw her consent at any time with no obligation to provide reasons. While informed consent was already given, the interviewer asked again permission for Claire being present and for the conversation being recorded.
The interview was then started with general questions about the interviewee’s disease, e.g., when it started, how it feels to live with it, etc. The interviewer then inquired after the different phases of the exam, and encouraged the interviewee to elaborate on particular answers in targeted follow-up question. In a second part of the interview, the content and conduct of the interview itself was discussed. The interviewer asked for feedback on particular elements of the interview, including its online format, spoken language, the duration, whether it was doable and the content.
Takeaway messages from the trial interview
1) General: the content and structure of the topic list allowed for an organic conversation and good quality data.
2) Discussion of the scanning procedure: It is good to differentiate between the four different phases of the MRI scanning procedure in the topic list as the different phases were experienced as (un)comfortable in different ways, and it is good to inquire after all phases separately so that the data we get about MRI does not gloss over important differences.
3) Research questions about first- and third person experiences (or objectification and alienation): doing the breathing exercises while lying down on a bed and being witnessed by others, and lying in the MRI on one’s stomach after receiving the bronchodilatator, both seem to be meaningful moments. It is thus good to be attentive when these elements are discussed.
4) Research questions about first- and third person experiences and cultural representations: while the subject expressed that it was interesting to see the produced MRI images at the end of the procedures, she also underlined that those images were just of lungs and not of disease. In addition, the interviewee expressed occasionally using an audio file (of a person experiencing breathlessness) to convey to others what it is like to have a lung disease, and so it is important to be mindful that meaningful representations of might also include non-visual artefacts.
5) Timing of the interview: It seems adequate to do a short questionnaire directly after all the procedures and then do the interview after some time went by. This would provide data about the feelings directly after the procedures and have something that we can reflect on in the interview later. Also it might be better to schedule more time for the interviews.
6) Language of the interview: It is best that the interviews will be in the participant’s mother tongue. Even though the interviewee was quite good at speaking English, when the interview moved to topics such as medication, scans, technology, medical conditions, anatomy, etc. it was sometimes a challenge to find the right words.
Credits: Irene Groenevelt (Tilburg University), Jenny Slatman (Tilburg University), Claire Barakat (Inserm)